2023
-

First year building The Bridge between clinical practice, software, and terminologies (OpenMRS + OCL + CIEL).
A globalized world demands ways to converge effort around a shared purpose. It makes me ask: what if we had a truly collaborative place to organize the health concepts the world relies on? What if we could speak a single clinical “code” and still be understood across systems, countries, and languages? Over the past year,…
-
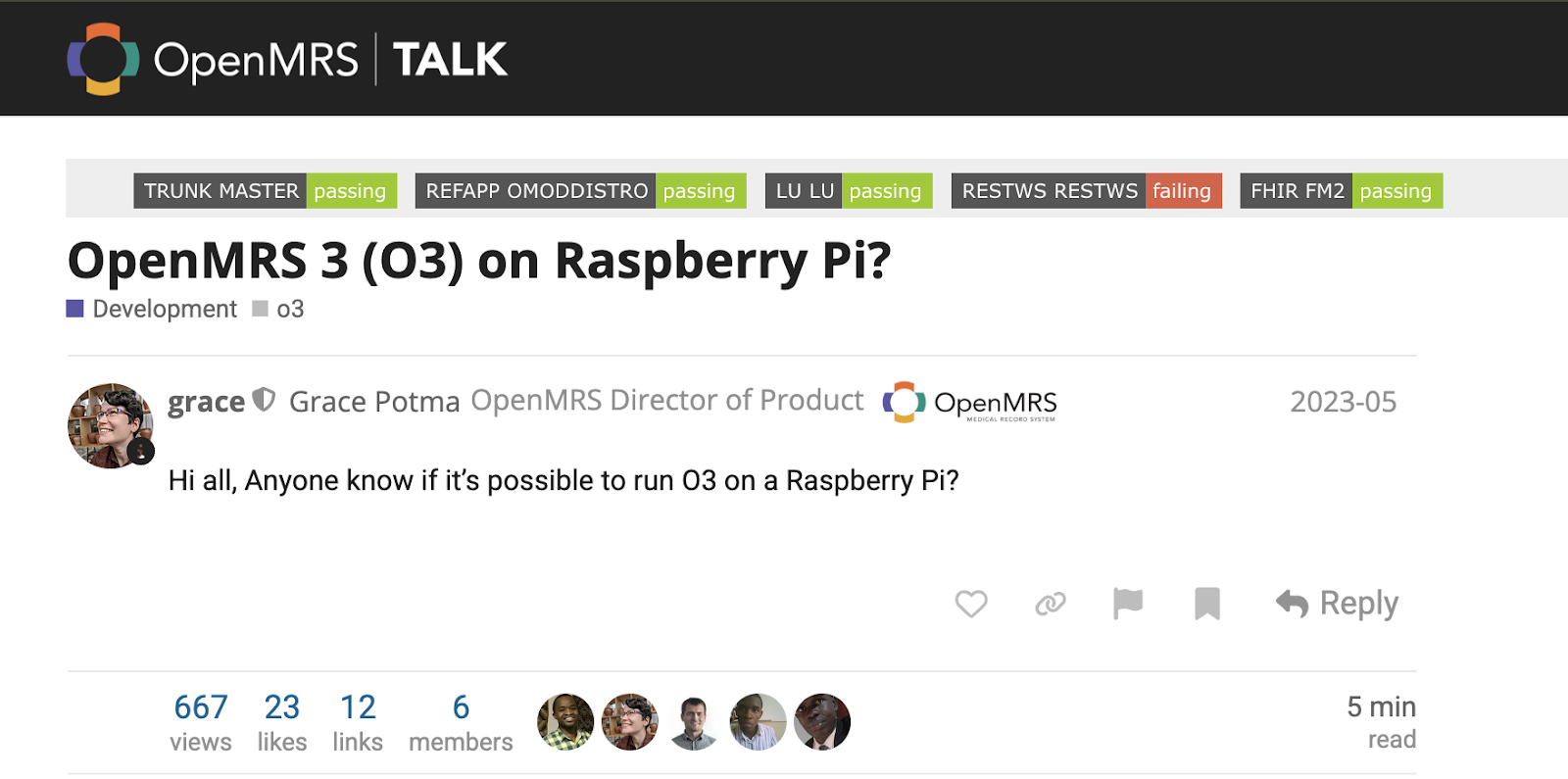
The EMR4All Journey: How a Volunteer-Led Mission is Transforming Digital Health in Remote Communities
EMR4All didn’t begin with funding or infrastructure. It began with the question: “Can we run OpenMRS on a Raspberry Pi?” Figure 1: The first question on Talk by Grace Potma about OpenMRS 3 (O3) on Raspberry Pi, which emphasised the vision of EMR4All in 2023 to become a reality. It was a technical curiosity that…
-
UgandaEMR+ O3 Success Story: UgandaEMR’s Journey with OpenMRS 3
UgandaEMR, an electronic medical record (EMR) system first released by the Ministry of Health (MoH) in July 2016, has become a widely used digital health solution across various healthcare facilities in Uganda. EMR systems like UgandaEMR have significantly improved patient management, clinical documentation and reporting throughout the country. In this article, you’ll learn how UgandaEMR…
